Study Overview
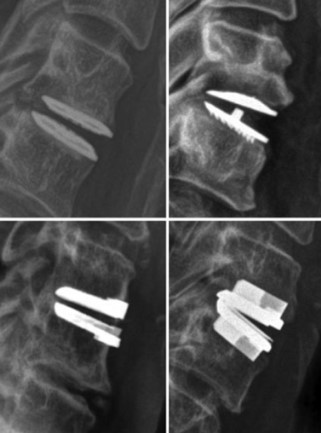
Cervical total disc replacement (CTDR) involves removal of the damaged cervical disc and replacement with an artificial device that is designed to maintain normal neck movement and relieve symptoms. While CTDR can help many patients, some patients experience problems over time such as recurring pain or discomfort in their neck and/or arms. Other patients may not experience any symptoms but develop radiographic changes seen on CT and X-ray imaging including bone loss, bone growth or changes to the device itself.
We don’t fully understand why these changes happen for some patients but not others. Through this study, we hope to find out; why some patients have poorer outcomes and/or device or bone related changes, how these changes progress and impact patients over time, and how we can better monitor and manage patients after CTDR?
We hypothesise that clinical factors such as age, gender, surgical level, features of the artificial disc and biological mechanisms including inflammation and bone growth may help explain why these changes happen.
This study aims to:
- Improve the process for routine clinical follow-up for patients undergoing CTDR
- Identify risk factors for recurring symptoms after CTDR
- Identify the range of radiographic changes and outcomes patients may develop after CTDR
- Understand the biological mechanisms of radiographic changes after CTDR
Study Progress and Update
Phase 1 – Preliminary Investigation. Our study first focused on a small group of patients with the M6-C™ artificial cervical disc as other studies and the Australian Therapeutic Goods Association guidelines suggest that these patients may experience radiographic changes. This study helped us to understand how common these changes are and which patients may be affected.
Phase 2 – Cohort Expansion. From our preliminary findings, we progressed to our second and current phase of the study which involves a larger group of patients with the M6-C™ artificial cervical disc to help us better understand these changes. We are continuing to monitor our patients for radiographic changes and are collecting blood and surgical samples to help us understand the biological mechanisms behind these changes.
Phase 3 – Recruitment of a New Cohort. After the completion of phase 2, we will start collecting the same information and samples from patients with CTDR devices other than the M6-C™ to further understand the role that CTDR devices and the way they function may play in these changes.
We are truly grateful to all patients who have participated so far. Your participation has allowed us to achieve some key milestones including:

If you would like to read more about our study findings so far, check out our peer reviewed publications below. Also see below some photos from the conferences we have attended to share the results of our work to other experts in the spine and spine surgery field.
- Häckel S, Gaff J, Pabbruwe M, Celenza A, Kern M, Taylor P, Miles A, Cunningham G. Heterotopic ossification, osteolysis and implant failure following cervical total disc replacement with the M6-C™ artificial disc. Eur Spine J. 2024 Mar;33(3):1292-1299. doi: 10.1007/s00586-024-08129-5. Epub 2024 Feb 16. PMID: 38363365.
- Celenza, A., Gaff, J., Pabbruwe, M. et al. The Total Disc Replacement Osteolysis Grading Scale – a simple, reliable and quantifiable tool for assessing, managing and reporting osteolysis after cervical total disc replacement. Eur Spine J (2025).
What’s Next?
We are in the early stages of our study and are actively enrolling eligible participants. Our goal is to reach full recruitment for phase 2 by mid-2026. Following this, we will begin to analyse genetic data from donated blood samples. This research may help us understand the outcomes of CTDR but also other spine-related conditions such as herniated discs and disc degeneration.
Invitation to New Participants
We are still enrolling eligible patients! If you or someone you know has a CTDR and has been or is currently a patient at Neurospine Institute, you may qualify to participate! To learn more, call us on 08 62858 520 or email us at research@nsiwa.com.au. Alternatively, if you would like to support our research with a donation, please visit our website here.
Thank You
To everyone who has joined or shown interest in our study – thank you! Your contribution is helping us advance research in spinal health and surgery. Together, we aim to develop better, personalised strategies for future patients.




